By GREGORY ZELLER //
Researchers have taken a big step forward in “bioprinting” – the science of fabricating living biological structures.
 Creating fully functional replacements for malfunctioning organs or missing limbs is still relegated to the science-fiction realm. But a team led by Michael Mak, an associate professor in the Department of Pharmacological Sciences in Stony Brook University’s Renaissance School of Medicine, has solved certain problems known to hamper bioprinting efforts – a potentially enormous advance for drug development, disease modeling and, someday soon, regenerative medicine.
Creating fully functional replacements for malfunctioning organs or missing limbs is still relegated to the science-fiction realm. But a team led by Michael Mak, an associate professor in the Department of Pharmacological Sciences in Stony Brook University’s Renaissance School of Medicine, has solved certain problems known to hamper bioprinting efforts – a potentially enormous advance for drug development, disease modeling and, someday soon, regenerative medicine.
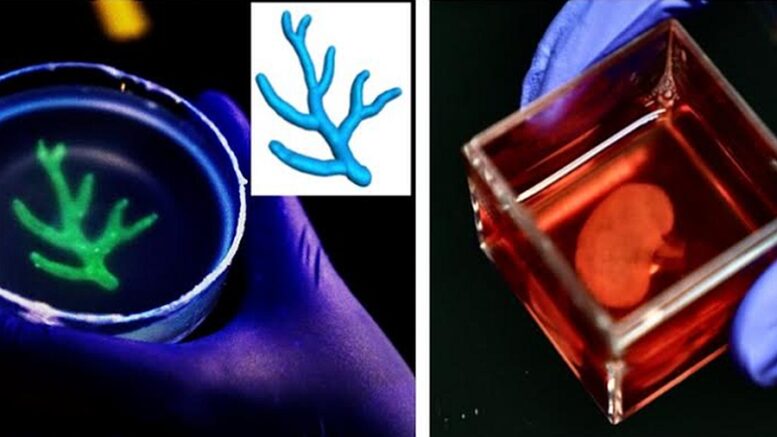
As detailed this month in a paper published by Nature Materials, a peer-reviewed scientific journal focused on materials science and engineering, Mak et al have developed a new method of bioprinting physiological materials known as TRACE (for Tunable Rapid Assembly of Collagenous Elements).

Michael Mak: Ink master.
This highly versatile biofabrication technique leverages collagen – the most abundant protein in the human body, and the key building block in vital organs and tissues such as skin, muscles and bones – to solve quandaries familiar to bioprinting.
Bioprinting aims to organize biochemicals, living cells and other biological materials into new bioengineered structures. Contemporary methods use “bioinks” and other biomaterials, along with computer-controlled 3D printing techniques, to construct living tissue models.
But achieving functionality in bioprinted tissues and organs has been challenging. Biological cells in traditional bioprinted tissues are unable to perform their natural activities in a host body, rendering most bioprinted structures unusable for clinical purposes and advanced medical applications.
Enter TRACE, which according to Mak – an expert in “mechanobiology,” extracellular matrixes and computational modeling, among other related disciplines – rectifies this problem by “using the body’s natural building blocks, particularly collagen, as bioinks.”
“Our method is essentially a novel platform technology that can be used to print wide-ranging tissue and organ types,” the scientist noted. “We figured out how to fabricate and manufacture complex user-designable tissue and organ structures … in a highly biocompatible manner and with direct incorporation of living cells.”
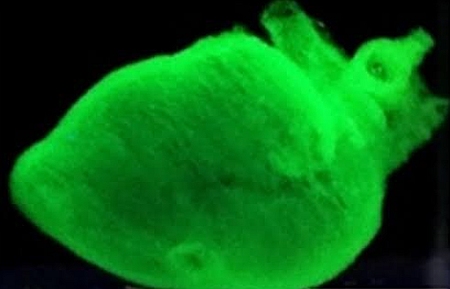
Size matters: TRACE can’t replace faulty organs, but it can bioprint functioning “mini” versions — like this “mini heart” — that are essential to laboratory research.
Collagen holds many tissues and organs in place – the body’s natural “glue” – and is key to directing cells to perform their myriad functions. This central role in the body’s physiological processes makes collagen a top bioink candidate, according to Mak, who was joined by nine co-authors on the new paper, “Instant Assembly of Collagen for Tissue Engineering and Bioprinting.”
The team details how TRACE rapidly accelerates collagen’s gelation process (essentially, converting a liquid to a solid). In this case, gelation is a matter of “macromolecular crowding,” in which inert materials such as proteins and nucleic acids are used to speed up the assembly reaction of collagen molecules.
This creates tissues composed of the same basic elements as those found inside the body – allowing TRACE to generate functional tissues and even “mini organs.”
While this does not equate to printing out a new human-sized heart or replacing a severed hand, the versatile biofabrication platform is a promising step toward achieving structural complexity and functionality – with advanced laboratory testing and other clinical applications in the offing.
“This work broadens the scope of controllable multiscale biofabrication for tissues across various organ systems,” Mak’s team notes in their study, “using collagen as a key component.”