By GREGORY ZELLER //
Applied DNA Sciences is ready to play a little monkey ball.
 The Stony Brook-based biotech – part supply-chain authenticator, part Polymerase Chain Reaction-based DNA technology leader, all innovation – is seeking New York State Department of Health approvals for its first monkeypox virus test.
The Stony Brook-based biotech – part supply-chain authenticator, part Polymerase Chain Reaction-based DNA technology leader, all innovation – is seeking New York State Department of Health approvals for its first monkeypox virus test.
Specifically, Applied DNA Clinical Labs – a wholly owned LLC launched in 2020 to flesh out the parent’s virology and oncology testing capabilities – has applied for the state validation package. It would be the spinoff’s second successful laboratory developed test this year, following NYS Health Department approvals in January for Applied DNA Clinical Labs’ next-generation COVID detector.
Named the Linea Monkeypox Virus 1.0 Assay, the PCR-based diagnostic is designed to identify the genetic signature of the monkeypox virus’ Clade II variant, currently the most prevalent U.S. strain.
Human monkeypox often presents as rashes, bumps or blisters on or around the genitals or on the hands, feet, chest or face, and usually causes flu-like symptoms including fever, headaches, chills and fatigue. The virus has historically proven fatal for about 11 percent of infected patients, according to the World Health Organization, though that case fatality rate has dropped between 3 percent and 6 percent in recent years.

James Hayward: Smarter than a pox.
Monkeypox vaccines exist. A vaccine once used against smallpox does the trick, (not surprising, since monkeypox is very similar to smallpox, which has been officially eradicated by successful global vaccinations: both are members of Orthopoxvirus, a genus of viruses in the family Poxviridae).
There’s also JYNNEOS, which was approved in 2019 for prevention of smallpox and monkeypox disease in adults, and earlier this month earned U.S. Food and Drug Administration emergency use authorization, increasing available doses for at-risk populations.
But vaccine supplies are still limited. And a fatality rate up to 6 percent is still a concerning number – especially with the challenge multiplied by a pandemic-weary society and a growing, misplaced stigma about this new viral threat.
Commonly misconstrued as a sexually transmitted disease – and mistakenly considered more of a risk for LGBTQ+ populations – human monkeypox can mimic common sexually transmitted infections in appearance but is not exclusively transmitted by sexual contact. Prolonged contact with infected clothing, bedding or linens can also spread the virus, as can hugging and kissing and contact with respiratory secretions.
That’s a lot of potential spreading – prioritizing quick detection and containment, according to Applied DNA Sciences President and CEO James Hayward.
“Our pursuit of monkeypox testing via LDT combines what we believe to be the quickest regulatory path for diagnostic approval with our proven ability to deliver short turnaround time-to-results,” Hayward said, adding those quick turnarounds, “as COVID-19 has taught us, can help contain virus spread.”
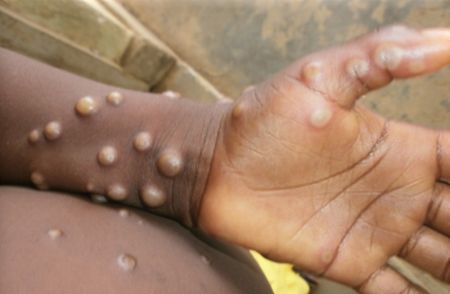
Blistering pace: Monkeypox is not exclusively sexually transmitted and can affect different parts of the body.
The monkeypox assay is part of Applied DNA Clinical Labs’ high-throughput disease-testing protocols, which focus on high-volume testing for infectious diseases. If the Department of Health greenlights the biotech’s latest LDT, monkeypox testing will be performed at the company’s molecular diagnostics laboratory in Stony Brook, “utilizing proven workflows to ensure accurate results and competitive turnaround times,” according to a company statement.
The progressive NYS Health Department application comes with confirmed monkeypox infections rising around the world. An ongoing outbreak of the viral disease was confirmed in May; the initial cluster was found in the United Kingdom, where the first cases were linked to an individual who’d traveled through Nigeria.
As of Monday, the global outbreak had exceeded 41,000 cases – including more than 14,000 U.S. cases – and resulted in 12 international deaths, including two in Spain and one in South America.
Applied DNA Clinical Labs’ assay also accompanies multiple monkeypox maneuvers by regional government and health officials. On July 20, Suffolk County Executive Steve Bellone announced Suffolk was receiving an additional 1,800 monkeypox vaccine doses from Albany, the county’s largest allotment to date; monkeypox vaccines are also being administered at the Nassau University Medical Center.
Meanwhile, both Stony Brook Medicine and Northwell Health have rolled out new monkeypox-vaccination efforts.
As the virus (and vaccinations) spread and testing becomes paramount, the Linea Monkeypox Virus 1.0 Assay should be ready to bring Applied DNA’s proprietary PCR-based diagnostics to bear, according to Hayward.
“Upon [state] approval, Applied DNA Clinical Labs has the testing capacity to deploy to help keep New Yorkers safe,” the CEO added. “Moreover, as the test kit manufacturer, we maximize the control over our own supply chain to ensure both quality and availability.”