By GREGORY ZELLER //
An international research consortium with Long Island roots may have identified the culprit behind COVID-19’s worst cases.
 It’s been well-documented that a large majority of patients who contract SARS-CoV-2, the virus that causes the COVID-19 disease, suffer only mild to moderate symptoms and fully recover.
It’s been well-documented that a large majority of patients who contract SARS-CoV-2, the virus that causes the COVID-19 disease, suffer only mild to moderate symptoms and fully recover.
Much more documented: The novel coronavirus can kill, with severe COVID-19 infections leading to Acute Respiratory Distress Syndrome, pulmonary inflammation, airway-blocking mucus secretions, blood clots and extensive lung damage – a system-wide breakdown that’s difficult to manage at best, and even with the assistance of invasive mechanical ventilators frequently turns fatal.
So, what’s the difference between the mild and severe cases? A group of topflight international medical-research organizations – including Cold Spring Harbor Laboratory, the Feinstein Institutes for Medical Research, Northwell Health, the Donald and Barbara Zucker School of Medicine at Hofstra/Northwell and others – may have the answer, revealed in a new paper published Thursday by Rockefeller University Press’ peer-reviewed Journal of Experimental Medicine.
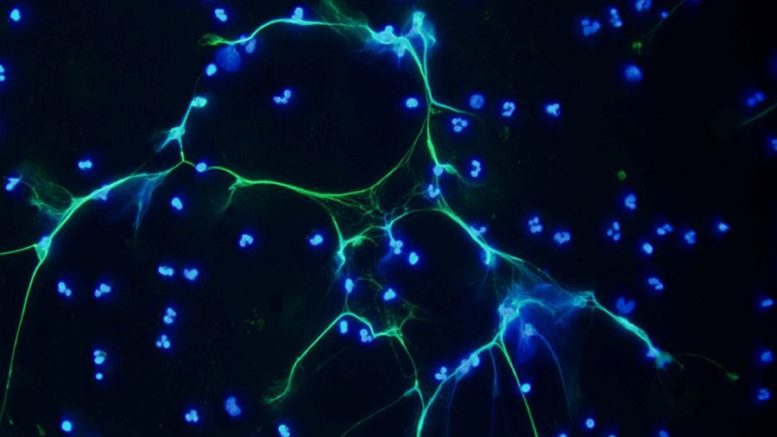
The paper (“Targeting Potential Drivers of COVID-19: Neutrophil Extracellular Traps”) explores whether overactive immune cells contribute to the most severe cases by producing Neutrophil Extracellular Traps, fiber networks that can snare and kill pathogens while protecting host cells.

Mikala Egeblad: NET gains.
In it, the international consortium – the “NETwork,” which also includes contributions from the University of California, the University of Michigan, the University of Utah, the University of Texas, McGill University Health Center in Montreal and the Centre Hospitalier Universitaire de Nancy in Laxou, France – suggests COVID-19 severity could be determined by neutrophils, overactive white blood cells dispatched by the body’s immune system to trap invading bacteria.
Those Neutrophil Extracellular Traps – basically tiny, sticky webs of toxic enzymes – are terrific for snagging and gobbling up invading pathogens. But they can also trigger Acute Respiratory Distress Syndrome and otherwise comprise pulmonary systems.
This apparent dichotomy represents a potentially giant leap toward understanding the methods of COVID-19’s madness, according to Cold Spring Harbor Laboratory biologist Mikala Egeblad, who pulled together the international consortium and acknowledges that the tangled science of NETs is fairly new.
“NETs were identified in 2004, but many scientists have never heard of them,” Egeblad, senior author of the new scientific paper, said Thursday. “Most of the researchers in the NETwork have worked on NETs in other diseases, and when we started hearing about the symptoms of the COVID-19 patients, it sounded familiar.”

Betsy Barnes: NET-level thinker.
Paper co-author Jonathan Spicer, a thoracic surgeon by trade and assistant professor of surgery at McGill University, said he and many Canadian colleagues also noticed similarities between the worst COVID-19 cases and other conditions exacerbated by Neutrophil Extracellular Traps.
“We see in these patients severe lung damage … caused by excess NETs and seen in cases of severe influenza,” Spicer said in a statement. “In addition, their airways are often clogged with thick mucus – and unlike most severe lung infections, these patients tend to form small clots throughout their body at much higher rates than normal.”
Recognizing the parallels between COVID-19 and the behaviors of other neutrophil-affected diseases could prove enormously beneficial to the legions of international scientists working to mitigate the global crisis, according to Betsy Barnes, a professor at Feinstein’s Institute of Molecular Medicine.
But there’s plenty of lab work still to come, noted Barnes, the paper’s lead and co-corresponding author.
“We propose that excess NETs may play a major role in the disease,” she said Thursday. “As samples from patients become available, it will be important to determine whether the presence of NETs associates with disease severity and/or particular clinical characteristics of COVID-19.”