By GREGORY ZELLER //
Northwell Health is trying on a little wearable technology.
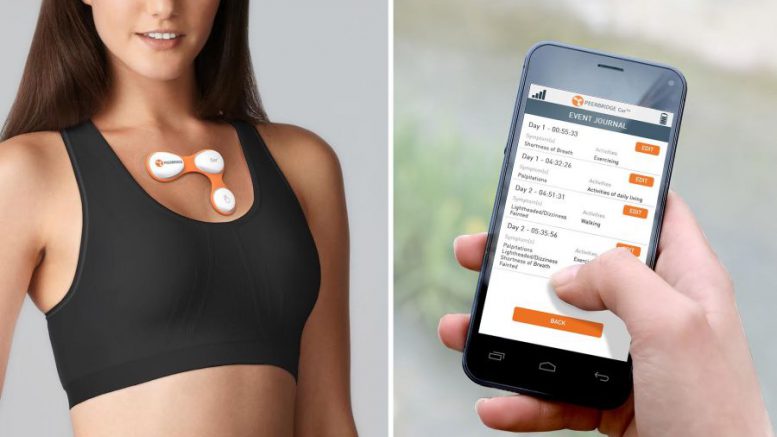
Make that a big wearable technology – as in, the Peerbridge Cor, a portable electrocardiogram monitor produced by Peerbridge Heath Inc.
 The device itself is small, but the “strategic partnership” with the New York City-based health-tech maker, announced Tuesday by the New Hyde Park-based health system, is anything but, with the Peerbridge Cor leading the way for other “medical-grade wearables” currently in development.
The device itself is small, but the “strategic partnership” with the New York City-based health-tech maker, announced Tuesday by the New Hyde Park-based health system, is anything but, with the Peerbridge Cor leading the way for other “medical-grade wearables” currently in development.
The move “solidifies Northwell’s commitment to improving the patient experience and the health of the communities it serves,” the health system said in a statement, while attempting to lower operational costs by employing state-of-the-art remote technologies that keep better track of patients’ conditions.
It’s a ginormous industry with virtually unlimited potential, and several Long Island companies – from Hauppauge-based product-design specialist Intelligent Product Solutions to Smithtown’s MIDI Medical Product Development – are already riding the wave of a wearable-medical-device market that could swell to $4.5 billion by 2020, according to worldwide business-intelligence aggregator Global Industry Analysts Inc.
Now Northwell Health is diving in. Wearable devices are already a big part of the neural-stimulation technologies championed by the Feinstein Institute for Medical Research, Northwell’s progressive research and development division, but in Peerbridge Health, the health system has made a new friend on the verge of wearable-tech breakthroughs in several critical areas, according to Northwell President and CEO Michael Dowling.
“Our collaboration with Peerbridge Health is part of our overarching strategy to introduce disruptive technologies that will help shape the future of healthcare delivery,” Dowling said Tuesday.
While Peerbridge Health boasts a pipeline of wireless-sensor patient-monitoring devices in various stages of readiness, the new partnership is centered primarily around the Peerbridge Cor. The compact, lightweight electrocardiography monitor tracks a patient’s pulse and heart rates, blood pressure, body temperature, respiratory status and other vital signs, and transmits all that data to mobile devices and the cloud for real-time analysis.
Peerbridge Health and Northwell Health recently completed a clinical trial designed to measure the Cor’s performance, comparing the portable electrocardiography device to other remote cardiovascular monitors in field conditions – and the Cor came up aces, according to Nicholas Skipitaris, chief of electrophysiology at Manhattan’s Lenox Hill Hospital, where the test run was based.
“The trial demonstrated this technology was superior to the current Holter monitor,” Skipitaris said in a statement, referencing the standard-bearer for remote cardiovascular monitoring.
“Not only did the Peerbridge Cor deliver excellent, multichannel, analyzable data, but it delivered less motion artifact and fewer false positives,” the MD added, noting the Cor excelled “despite our patient’s active New York lifestyle.”
Providing accurate, actionable medical data in real-world conditions is the big picture at Peerbridge Health, according to CEO Arthur Bertolero, who suggested Northwell Health – New York State’s busiest healthcare provider and largest private employer, by numbers of patients and employees, respectively – was the perfect partner to help bring that mission to fruition.
“Our vision is to help consumers partner with their physicians through easily accessible remote-monitoring options,” Bertolero said. “Northwell has been a strong strategic partner, sharing our passion to bring effective digital solutions to consumers and health professionals with the highest medical standard.”
Although it is not yet commercially available, the patent-pending Peerbridge Cor is expected to receive FDA approval and hit the market later this year.
It can’t happen fast enough, according to Northwell Health, which notes American Heart Association statistics citing cardiovascular disease and stroke as the top two killers of American adults – and U.S. Centers for Disease Control & Prevention projections that annual costs associated with cardiovascular disease will surpass $818 billion by 2030.
To curb those expenses and improve patient outcomes, “cardiovascular monitoring, both inside and outside medical environments, is critical,” Northwell Health said.