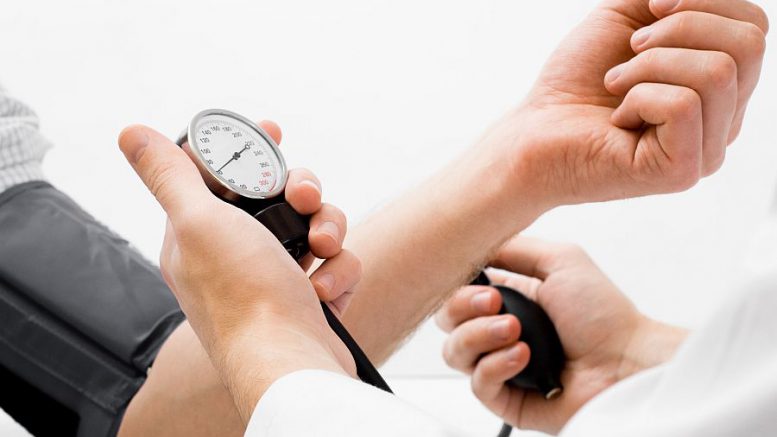
A new study by the head of the Feinstein Institute for Medical Research may open the door to bioelectronic treatments for hypertension and high blood pressure.
In a study published this month by the prestigious peer-reviewed scientific journal Nature Biotechnology, researchers from the Feinstein Institute – the R&D arm of the Northwell Health system – and the Karolinska Institutet, Sweden’s top medical university, discuss findings regarding T-cells and their ability to produce neurotransmitters that can regulate blood pressure.
T-cells are white blood cells and part of the immune system. They contain the enzyme choline acetyltransferase (it goes by ChAT), which produces the neurotransmitter acetylcholine, and have been shown to respond to electronic stimulation of the vagus nerve.
Nerve-stimulation technologies are one of the research pillars at the Feinstein Institute, where President and CEO Kevin Tracey is widely regarded as a global bioelectronics pioneer. The institute is also home base for Sanguistat, a nascent startup and the commercial face of the Neural Tourniquet, an electrical nerve-stimulation device for staunching blood loss, based on Tracey’s research.
While previous studies showed promising effects on “neural mechanisms” accessed bioelectronically through the spleen, the joint project with the Stockholm-based Karolinska Institutet “reached deeper into the smaller arteries,” noted Tracey, one of the study’s lead authors, and “we found what we were looking for.”
Working off the findings of a previous study published by the Feinstein Institute in a 2011 issue of the journal Science, showing T-cells in the spleen can affect immune functions controlled by the vagus nerve, researchers focused on T-cells that synthesize the ChAT acetylcholine.
The study – which received financial support from the National Institutes of Health and the National Institute of General Medicine – determined that mice without ChAT in their T-cells exhibited higher blood pressure and concluded the T-cells were the source of the arterial pressure-easing neurotransmitter.
“We were looking to identify triggers … to aid with conditions such as high blood pressure,” Tracey said in a statement, adding scientists will now look to leverage their findings into “new therapies that will improve the lives of millions.”
Specifically, now that they’ve identified ChAT’s role in blood pressure regulation, researchers will explore potential bioelectronic medicine treatments for high BP and hypertension – promising news for the 33 percent of Americans whose pressure runs high.
Paper co-author Peder Olofsson, a Karolinska Institute PhD who worked at the Feinstein Institute from 2009 to 2015 and has since returned to Karolinska as founding director of its Center for Bioelectronic Medicine, said the joint study – one of many between the Swedish university and the Feinstein Institute over a decade-long partnership – underscored the importance of “interdisciplinary collaborations in bioelectronic medicine.”
Such collaborations “offer an opportunity to improve understanding of little-understood physiological mechanisms,” Olofsson noted. “This study … represents one such advance.”
The Karolinska Institutet, home of the committee that selects winners of the Nobel Prize in physiology or medicine, made a bioelectronic splash in 2015 when researchers there built a fully functional neuron – capable of mimicking human nerve cells – using organic bioelectronic components.
Tracey and Olofsson are likely to discuss these and other biolectronic findings this week when they share keynote-address duties at Key Symposium 2016: Bioelectronic Medicine – Technology Targeting Molecular Mechanisms, scheduled to run Wednesday to Friday at the New York Academy of Sciences.