By GREGORY ZELLER //
A biotech with a penchant for leather, foreign automobiles, high-tech weaponry and next-gen digital imaging is finally getting around to pharmaceuticals.
Actually, that’s not true – Stony Brook-based Applied DNA Sciences has already this year announced multiple pharma-nutra-focused forays, including the production of DNA chains for in-vitro diagnostic and DNA-based therapeutic research purposes.
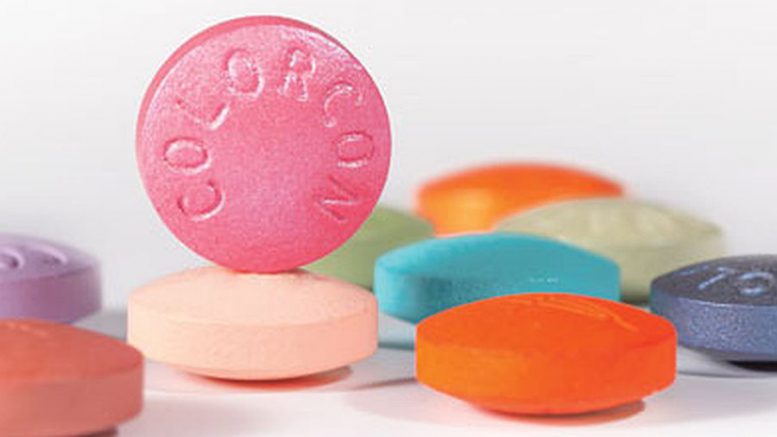
 But in announcing a technology license memorandum of understanding with Pennsylvania-based “hard dose” specialist Colorcon Inc., Applied DNA may have taken its boldest step yet into another potentially massive vertical: the molecular tagging and tracking of ingestible medications.
But in announcing a technology license memorandum of understanding with Pennsylvania-based “hard dose” specialist Colorcon Inc., Applied DNA may have taken its boldest step yet into another potentially massive vertical: the molecular tagging and tracking of ingestible medications.
Colorcon is a major manufacturer of pills, capsules and other physical forms of medication, including product coatings. The technology-license agreement combines the producer’s portfolio of film-coating systems, inks and colors with Applied DNA’s proprietary molecular taggants in an effort to “significantly reduce the risks associated with counterfeit and falsified medications entering the drug supply chain,” according to the Stony Brook biotech.
The Applied DNA tech offers an important market advantage for a major manufacturer with a worldwide network of technical-service laboratories, a global customer base and a product catalogue ranging from excipients (ingredients that “bulk up” medicinal pills) to confectionary inks to hydrophilic (and other “controlled-release”) tablets, according to General Manager Kelly Boyer, who heads Colorcon’s film-coating operations.
“By incorporating molecular taggants from Applied DNA in our specialty excipients, Colorcon is able to offer a simple and seamless solution to our pharmaceutical and nutraceutical customers to address counterfeiting and product-diversion issues,” Boyer said Monday.
 The non-binding MOU, which is expected to be followed by a definitive agreement, also creates another commercial platform for Applied DNA’s unbreakable, eminently traceable DNA-based authentication technologies, following recent thrusts into the Asian/West African fertilizer trade, global plastics production and even the U.S. Army’s bio-warfare research programs, among others.
The non-binding MOU, which is expected to be followed by a definitive agreement, also creates another commercial platform for Applied DNA’s unbreakable, eminently traceable DNA-based authentication technologies, following recent thrusts into the Asian/West African fertilizer trade, global plastics production and even the U.S. Army’s bio-warfare research programs, among others.
Applied DNA President and CEO James Hayward called the tech-licensing deal “the culmination of a multi-year effort” focused on the pharma-nutra industries, where manufacturers “are moving quickly to address supply-chain concerns and comply with federal serialization mandates across multiple continents.”
“In partnering with Colorcon, we are aligned with a truly innovative, high-quality science and technology company in bringing our novel technology to the center of the pharmaceutical industry,” Hayward said. “The result will be end-to-end protection of a significant portion of the total addressable pharmaceutic market.”
That’s “especially important” in the United States, the CEO added, noting domestic pharmaceuticals account for about 45 percent of the global market.
Bob Miglani, Applied DNA’s business-development chief, called the MOU a “breakthrough opportunity” for the Stony Brook biotech to gather critical data “gathered from authentication of the dosage forms themselves.”
“By better understanding trade flows and vulnerabilities in a complex global supply chain, we are helping pharmaceutical leaders make better decisions on managing distribution patterns, frequent monitoring and deploying preventative measures for their products around the world,” Miglani said in a statement.