By GREGORY ZELLER //
Marking a national first, Northwell Health on Monday delivered the first set of COVID-19 vaccination booster shots to organ-transplant patients.
![]() The Feinstein Institutes for Medical Research, Northwell’s Manhasset-based R&D mecca, delivered the shots as part of a nationwide Moderna clinical trial exploring the efficacy of boosters in patients living with a transplanted liver or kidney.
The Feinstein Institutes for Medical Research, Northwell’s Manhasset-based R&D mecca, delivered the shots as part of a nationwide Moderna clinical trial exploring the efficacy of boosters in patients living with a transplanted liver or kidney.
Transplant patients run a high risk of contracting COVID-19 and becoming severely ill, since immunosuppressive drugs common to transplant cases may fritz their ability to build a strong immune defense against the virus – even after receiving their first vaccine doses.

Lewis Teperman: Evidence-based.
“We are eager to provide [organ-transplant recipients] more vaccine to help protect them, and to gain much-needed scientific evidence to help doctors best treat their patients,” noted principal investigator Lewis Teperman, Northwell Health’s director of transplant services.
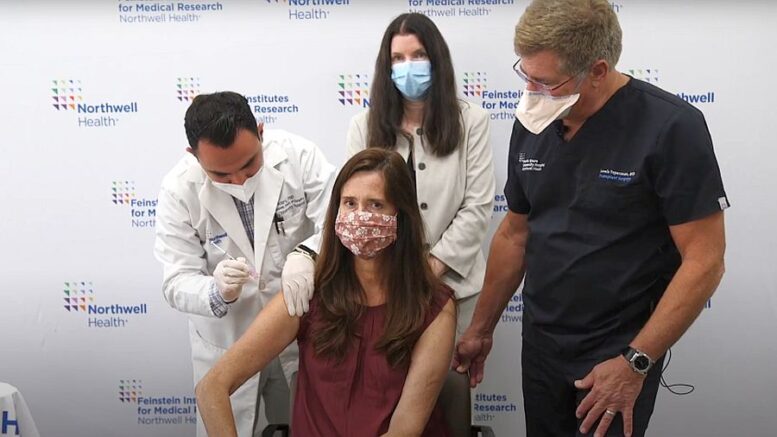
The trial started with George W. Hewlett High School teacher Darla Smyth, who received her liver transplant 28 years ago and was the first of three transplant patients receiving their booster shot Monday. Researchers will monitor boosted patients’ immune antibody responses for 28 days.
“When you weigh the options – either get sick or get vaccinated again – my choice was clear,” Smyth said Monday. “I am hoping this third shot will help give me some lasting protection from this virus.”
Moderna is planning to enroll 240 participants – including healthy volunteers and patients who have received a solid organ transplant within the last six months – in the booster trial, which is taking place at the Feinstein Institutes and six other national sites.
The trial is the latest of several COVID-vaccine studies initiated at the Feinstein Institutes. In March 2020, the Northwell facility announced its first set of clinical trials focused on the safety and efficacy of potential COVID-19 therapies. Since then, the Feinstein Institutes has initiated nearly 20 COVID-focused clinical trials and programs; earlier this month, it announced a National Institutes of Health-funded booster-shot study focused on patients with autoimmune diseases.
All told, the Feinstein Institutes has enrolled more than 1,800 patients in COVID-vaccination studies, fueling 500-plus peer-reviewed scientific publications related to the virus.
“Since the start of the COVID-19 pandemic, the Feinstein Institutes focused on conducting clinical trials in an effort to find safe and effective treatments,” Christina Brennan, the Feinstein Institutes’ vice president of clinical research, said in a statement. “Now, with the support of Moderna, we hope this vaccine clinical trial will give insight into the best way to protect a high-risk population.”