By GREGORY ZELLER //
Stony Brook University researchers are reporting significant gains in the war on cancer – specifically, an anticancer drug showing real potential to reduce cancerous tumors.
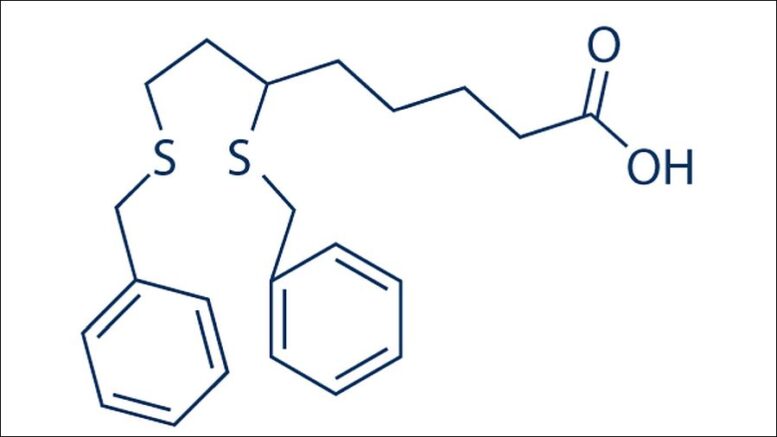
 That’s the word on CPI-613, the lead agent in a class of anticancer compounds currently undergoing U.S. Food and Drug Administration clinical trials – and already proving effective in reducing the spread of most carcinoma cell lines, a common type of cancer that starts in skin cells or the tissue lining kidneys and other vital organs.
That’s the word on CPI-613, the lead agent in a class of anticancer compounds currently undergoing U.S. Food and Drug Administration clinical trials – and already proving effective in reducing the spread of most carcinoma cell lines, a common type of cancer that starts in skin cells or the tissue lining kidneys and other vital organs.
The lead agent and other anticancer compounds were developed decades ago by Stony Brook Department of Biochemistry and Cell Biology Associate Professor Paul Bingham and Zuzana Zachar, now a research assistant professor in the university’s Institute for STEM Education.
With the drug finally running the clinical gauntlet, Bingham, Zachar (who also directs SBU’s Master in Teaching Science Program) and five co-authors are sharing research that not only shows the novel compound’s standalone success but suggests CPI-613 – used in combination with other cancer treatments – “could have efficacy [in] reducing some tumors.”

Paul Bingham: “Potentially powerful” weapon.
Their findings, published this week in the peer-reviewed scientific journal PLOS ONE, position CPI-613 as “potentially powerful for general and broad-based clinical approaches” to battling cancer, Bingham said Wednesday.
“Our new preclinical research shows that the anticancer mechanisms we originally reported with CPI-613 remain intact against most cancer cell lines,” the professor noted. “Secondly, we also show that failures to respond to CPI-613 in clinical applications have a very simple, specific, tumor-general cause.”
While CPI-613 is very good at fritzing the metabolic cycle that spreads cancer, certain transmission pathways still exist, allowing infected electrons to work around the CPI-613 blockade. Bingham, Zachar et al were able to identify that baleful back door: a fatty acid beta-oxidation process that, it turns out, is very friendly to mitochondrial energy metabolism.
By targeting this sinister alternative electron flow – what Bingham called a “fatty acid bypass” – clinicians can “substantially enhance the anticancer performance of CPI-613,” according to SBU.
New Jersey-based Cornerstone Pharmaceuticals (formerly Rafael Pharmaceuticals) initially licensed the CPI-613 technology from The Research Foundation of the State University of New York in 2001 and first brought the drug – which the company calls Devimstat – to pre-clinical testing in 2011.
As the exclusive licensee, and with the FDA’s blessing, Cornerstone is now conducting the extended CPI-613 clinical trials.
To date, those trials have shown that tumor reduction occurs in “only some patients,” according to SBU – but researchers are hopeful the ongoing testing will show how to use the anticancer agent more effectively in combination with existing cancer treatments, “especially with difficult-to-treat tumors.”