By BRAD CARLSON //
We’ve entered the Age of Neuromodulation.
 The use of neuromodulation devices – which electrically stimulate nerves to suppress symptoms or reinvigorate poor function – has grown rapidly over the last 20 years, and the applications just keep expanding
The use of neuromodulation devices – which electrically stimulate nerves to suppress symptoms or reinvigorate poor function – has grown rapidly over the last 20 years, and the applications just keep expanding
Though it was not technically an NMD, the 1960 invention of the implanted pacemaker paved the way for modern NMDs by establishing the basic technology: implanted, battery-powered electronics delivering electrical impulses directly to human tissue through a tiny wire.
Today, NMDs are common – some implanted in the body, directly stimulating the brain or peripheral nerves, some worn outside the body, stimulating nerves close to the skin’s surface.
It’s not quite a head-to-toe thing – there are no NMDs for toes, yet – but it’s definitely head-to-lower extremities, as neuromodulation targets everything from Parkinson’s disease to phantom pain syndrome.
One of the largest current applications is a spinal cord stimulator implantation for treating chronic pain. The device, implanted in the back, stimulates spinal-cord nerves to block pain signals from reaching the brain. The SCS implant was invented in 1967; today, roughly 50,000 devices are implanted each year.

Brad Carlson: A-OK with NMDs.
While NMDs are broadly effective, they’re not 100 percent guaranteed. Due to as-yet-unknown factors, some devices fail to work for some patients – and may even worsen symptoms, though this is unlikely.
One example of this dichotomy is the vagus nerve stimulator developed by London-based medical device maker LivaNova to treat drug-resistant epilepsy.
The VNS is implanted in the chest with a lead connected to the left vagus nerve in the neck. It delivers electrical pulses that follow the nerve to the brain, aiming to disrupt epileptic seizures, and has proven to be highly effective in many patients.
But not every patient. And there can be undesirable side effects, such as voice impairment. The device connects to the vagus near the laryngeal nerve, and the electric pulses sometimes make it difficult for patients to speak.
I led the research and development team for LivaNova for several years, and a significant focus of our work was improving VNS effectiveness for a higher percentage of patients. Fortunately, the VNS device stimulates for only 30 seconds or so every five minutes, so the speech impairment is temporary – and the device was largely effective, sometimes highly effective, when it came to delivering relief from seizures.
Vagus nerve stimulation is also approved for the treatment of major depressive disorder, and research is ongoing to investigate VNS for several other diseases and conditions. It’s a central focus at the Manhasset-based Feinstein Institutes for Medical Research, where a broad range of indications are routinely considered.
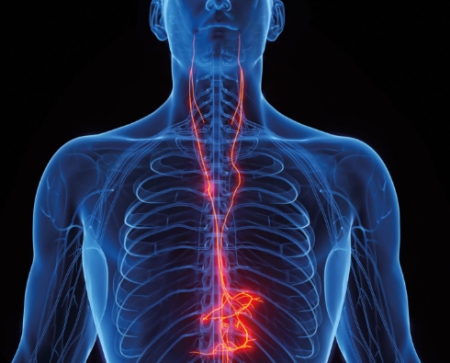
Vagus odds: There’s a great chance the all-important vagus nerve will play a huge part in future medical care.
The full extent of our nervous system’s behavior is not fully understood, leaving much opportunity for the discovery of new NMD applications. Consider sleep apnea, another rapidly growing neuromodulation space.
You’ve probably seen the television commercials by Inspire Medical Systems, urging the apnea-diagnosed to exchange their breathing masks for implanted devices. This device stimulates the hypoglossal nerve to reposition the tongue out of the airway during sleep.
Unlike spinal cord and vagus nerve implants, which target sensory nerve fibers, the sleep apnea device affects motor nerve fibers, exercising tongue muscles to keep the airway clear. More companies, including LivaNova, are bringing similar sleep apnea devices to market soon.
There’s also been plenty of progress with those outside-the-body, transcutaneous NMDs. Manhasset-based brain-computer interface pioneer Neuvotion, a startup spun out of the Feinstein Institutes, is developing wearable technology that can stimulate motor nerve fibers in the lower arm to facilitate motion in paralyzed patients.
They’ve leveraged Hauppauge-based Intelligent Product Solutions to help develop the hardware and software. Worn on the forearm and thumb, the Neuvotion device will ultimately enable grasping of objects such as drinking cups, restoring both function and independence.

Getting Neumotion-al: Feinstein Institutes spinoff Neumotion intends to restore both mobility and quality of life.
Other transcutaneous NMDs are in development specifically for the treatment of opioid addiction.
Devices are also getting smarter. Some, like the Inspire Medical implant, use sensors to optimize stimulator performance – rather than continuously stimulating, they read conditions to determine when stimulation is needed, preserving battery life.
Some implants also use rechargeable batteries, extending time between replacement surgeries (though this can require the patient to maintain the charge, or risk malfunction).
Implanted NMDs are critical for treating some diseases, but they are invasive, require surgeries and are permanent. Special consideration must be engineered into the device to make it compatible with common procedures like MRIs. And even those rechargeable batteries have limited lifetimes.
Fortunately, engineers have overcome many of these limitations – you and your NMD can survive a CT scan, if you’re careful – and continue to improve device capabilities for improved patient outcomes.
As new neuromodulation devices gain regulatory approval and proliferate the market, the potential grows for managing debilitating conditions and improving quality of life for many different people. Researchers and engineers will continue to build their understanding of disease and injury conditions – and develop new NMDs that can address them effectively.
Brad Carlson is vice president of technology and business development at Hauppauge-based Intelligent Product Solutions.







