By TERRY LYNAM //
My mother has been dead for almost three years, but I still think of her often, particularly when I read the steady stream of new developments in Alzheimer’s research – glimmers of hope to the more than 6.5 million Americans living with the disease.
 Remarkably, my mom lived to age 90, after being diagnosed with Alzheimer’s in her late 70s. But her cognitive functions declined and she suffered from many physical ailments during her final dozen years or so.
Remarkably, my mom lived to age 90, after being diagnosed with Alzheimer’s in her late 70s. But her cognitive functions declined and she suffered from many physical ailments during her final dozen years or so.
In the end, she was bedridden, incontinent and in excruciating pain, struggling to converse with loved ones (though she never lost the ability to recognize them).
She’d been diagnosed after a series of troubling incidents – hoarding groceries, failing to take her prescription meds, forgetting conversations from just minutes earlier. Once, she got lost driving home in an area where she’d lived for decades.
We ended up having a family intervention: taking away her car, moving her out of the Florida home where she’d lived for more than 30 years, relocating her to an assisted-living facility in Colorado, near my sister.
“Your father would turn over in his grave if he knew what you were doing to me,” she told me, as we moved her into the facility.
Countless others with aging parents suffering from dementia can share similar stories of heartbreak and guilt. Others may wonder if they’ll face a similar fate.

Terry Lynam: Mind games.
There’s hope on the horizon. Sometime before July 6, the U.S. Food & Drug Administration will decide whether to grant full approval to the first brain plaque-busting drug shown to be effective in reducing cognitive decline in Alzheimer’s patients.
Earlier this month, a six-member FDA advisory panel unanimously endorsed the benefits of Lecanemab, clearing the way for the FDA to consider final approvals.
It’s not a cure for Alzheimer’s. But the drug – sold under the brand name Leqembi and developed jointly by Japanese pharmaceutical company Eisai and Massachusetts-based biotech Biogen – already received accelerated FDA approval in January for people with mild cognitive impairment or mild dementia, based on trial results showing it slowed cognitive decline by 27 percent.
If it’s ultimately approved, the questions become: Who gets it, how quickly, and how much?
Centers for Medicare and Medicaid Services Administrator Chiquita Brooks-LaSure said in a June 1 statement that CMS will ensure that anyone with Medicare Part B who meets the necessary criteria will be covered, “if the FDA grants traditional approval.”
Meeting the criteria, however, is the tricky part. That would require patients, physicians and clinical teams to participate in “the collection of evidence about how these drugs work in the real world,” according to Brooks-LaSure, “also known as a registry.”
Without that, out-of-pocket costs could run as high as $26,500 a year, according to the CMS.
Lecanemab would become only the second Alzheimer’s disease treatment to be approved in the United States since 2002. In a controversial 2021 decision, the FDA granted accelerated approvals for Aducanumab (sold under the brand name Aduhelm), which is also produced by Eisai and Biogen and also targets amyloid plaques in the brain.
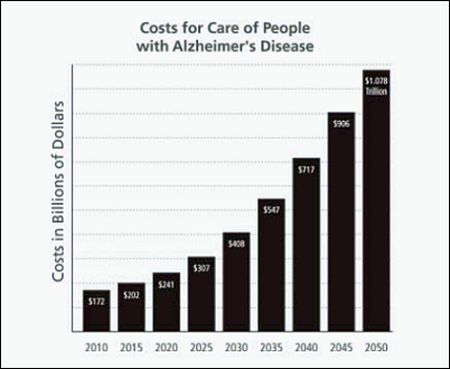
Source: BrainFacts.org
Following Aducanumab’s approval, three scientists from an independent FDA advisory committee resigned in protest. After an 18-month congressional investigation, a December 2022 report found the FDA’s approval process “rife with irregularities” – and strongly criticized Biogen for planning to set “an unjustifiably high price” for Aducanumab ($56,000 a year).
Given the controversy surrounding Aducanumab and its unclear clinical benefits, the FDA has yet to give the drug its full approvals – and has limited reimbursements for using it.
The outlook for Lecanemab appears promising, but the long-term efficacy of drugs that remove plaque from the brain (known as monoclonal antibodies) continues to be disputed within the medical and scientific communities. There are numerous FDA-approved drugs that treat Alzheimer’s symptoms related to memory and behavior – the FDA gave supplemental approval in May for a drug to treat agitation) – but a momentous breakthrough to treat the underlying disease has been elusive.
Meanwhile, citing high costs and limited benefits, some argue that treating Alzheimer’s is too costly and the focus should be on prevention. A 2020 report in The Lancet identified 12 modifiable risk factors that account for about 40 percent of the world’s dementia cases (estimated at 55 million by the World Health Organization), including lifestyle choices (smoking, excessive alcohol use, obesity), medical conditions (hypertension, hearing impairments, depression, diabetes) and environmental factors (air pollution chief among them).
Health experts agree it’s never too early (or too late) for preventive action – Alzheimer’s is not inevitable. And maybe one day they will find that miracle cure.
Until then, healthcare providers, policymakers and pharmaceutical companies that have been profiting so handsomely should put a greater focus on public education – and more resources into preserving brain cells while they’re still healthy.
Terry Lynam is a communications consultant and former senior vice president/chief public relations officer for Northwell Health.